
Read this K-value off the chart (approximately 21.3). Note where the line crosses the methane axis.Connect the points with a straight line.
DEPRIESTER CHART FOR HYDROCARBONS HOW TO
On the right-hand vertical axis, locate and mark the point containing the temperature 60☏. Uses an interactive simulation to explain how to use a DePriester chart for hydrocarbons to determine K values and show how K values change with temperature.Part 2: A refinery in Northwest Pennsylvania produces asphalt as an important product that brings revenue, particularly during the summer months. liquid equilibrium ratio and the enthalpy of hydrocarbon mixtures con. On the left-hand vertical axis, locate and mark the point containing the pressure 100 psia. K values for light hydrocarbons at high temperatures (DePriester Chart).Example įor example, to find the K value of methane at 100 psia and 60 ☏. Many DePriester charts have been printed for simple hydrocarbons. "K" values, representing the tendency of a given chemical species to partition itself preferentially between liquid and vapor phases, are plotted in between. These nomograms have two vertical coordinates, one for pressure, and another for temperature. DePriester in an article in Chemical Engineering Progress in 1953.

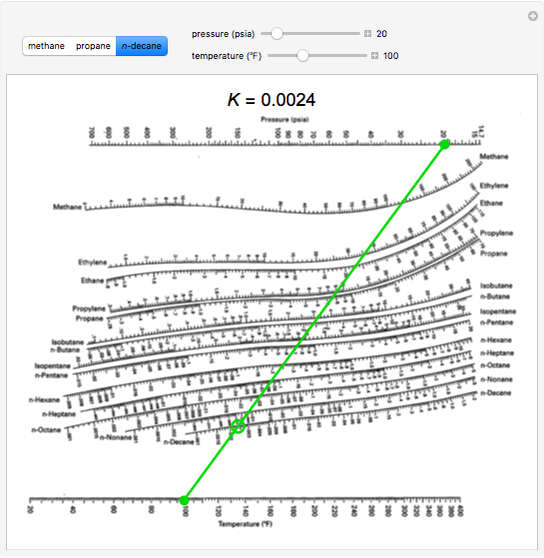
DePriester Charts provide an efficient method to find the vapor-liquid equilibrium ratios for different substances at different conditions of pressure and temperature.
